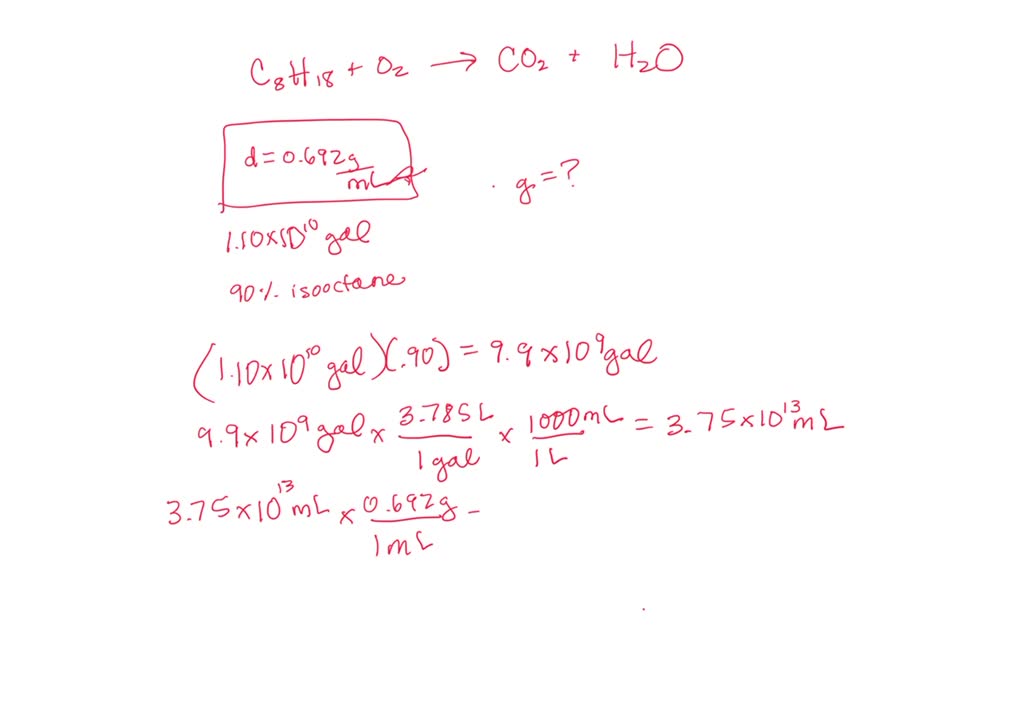
SOLVED: Assuming gasoline is 90.0% isooctane, with a density of 0.692 g/mL, what is the theoretical yield (in grams) of CO2 produced by the combustion of 1.10 x 1010 gallons of gasoline (

Amazon.com: SE Survivor Series Collapsible Water Carrier 20L (5 Gallon) - OD-WC205P : Sports & Outdoors