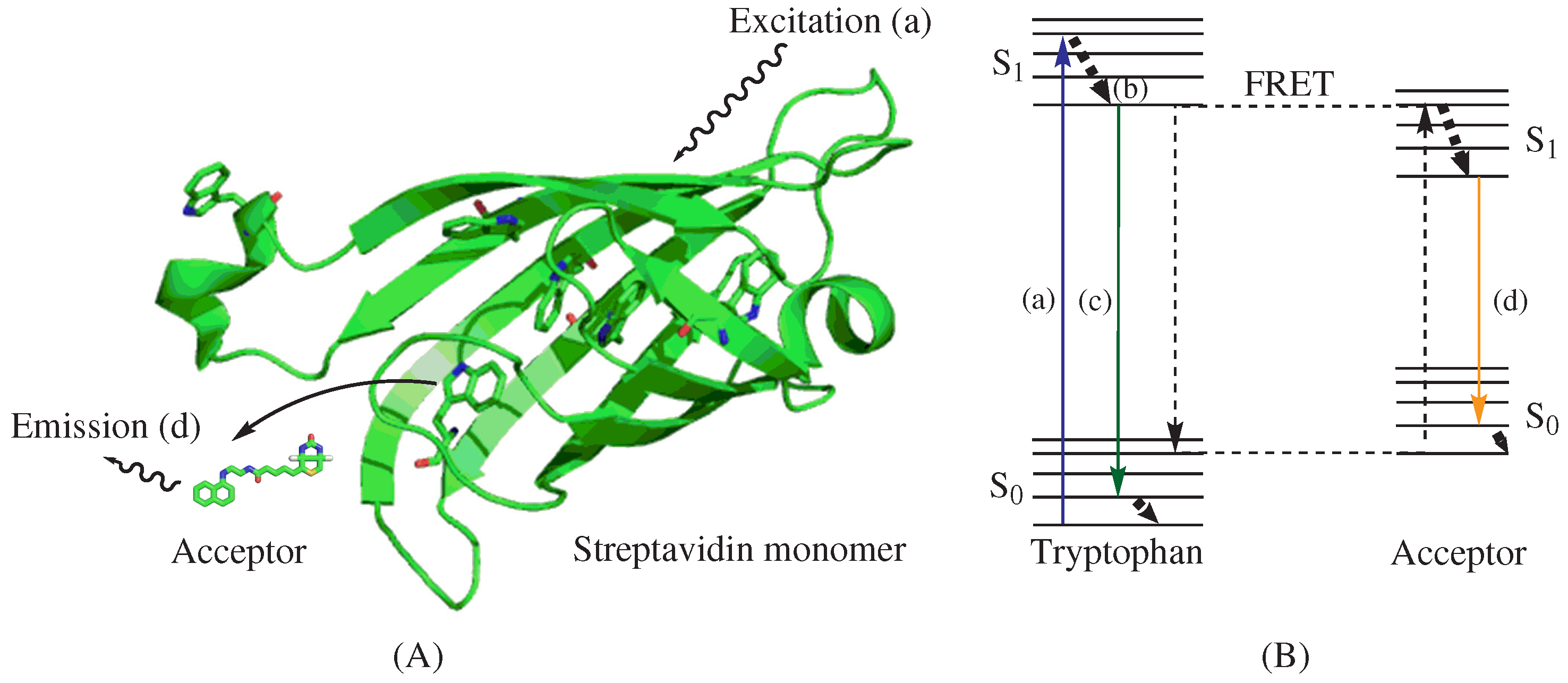
![PDF] Using tryptophan fluorescence to measure the stability of membrane proteins folded in liposomes. | Semantic Scholar PDF] Using tryptophan fluorescence to measure the stability of membrane proteins folded in liposomes. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/757f28eef910ecefa42ba89d6a924beedc4b2715/4-Figure6.1-1.png)
PDF] Using tryptophan fluorescence to measure the stability of membrane proteins folded in liposomes. | Semantic Scholar

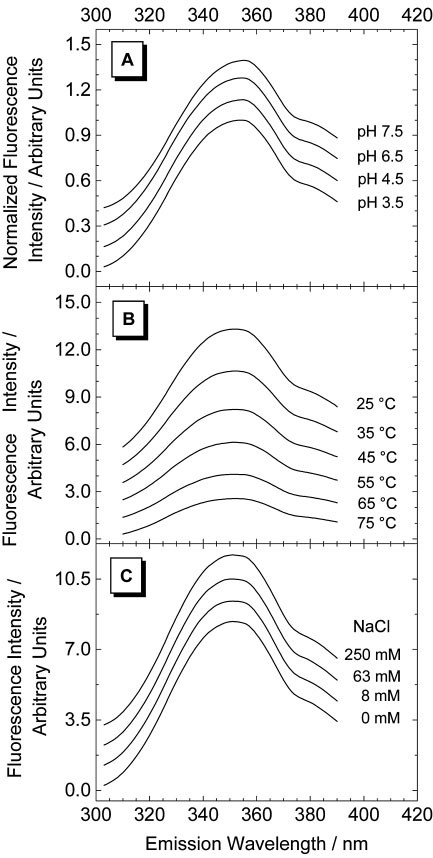
Spectra of tryptophan fluorescence are the result of co-existence of certain most abundant stabilized excited state and certain most abundant destabilized excited state - ScienceDirect
![PDF] Selenomethionine Quenching of Tryptophan Fluorescence Provides a Simple Probe of Protein Structure. | Semantic Scholar PDF] Selenomethionine Quenching of Tryptophan Fluorescence Provides a Simple Probe of Protein Structure. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a194744fecdf4f5fd146310e0e1121ca422b5a5f/21-Figure1-1.png)
PDF] Selenomethionine Quenching of Tryptophan Fluorescence Provides a Simple Probe of Protein Structure. | Semantic Scholar
Quenching of tryptophan fluorescence in a highly scattering solution: Insights on protein localization in a lung surfactant formulation | PLOS ONE

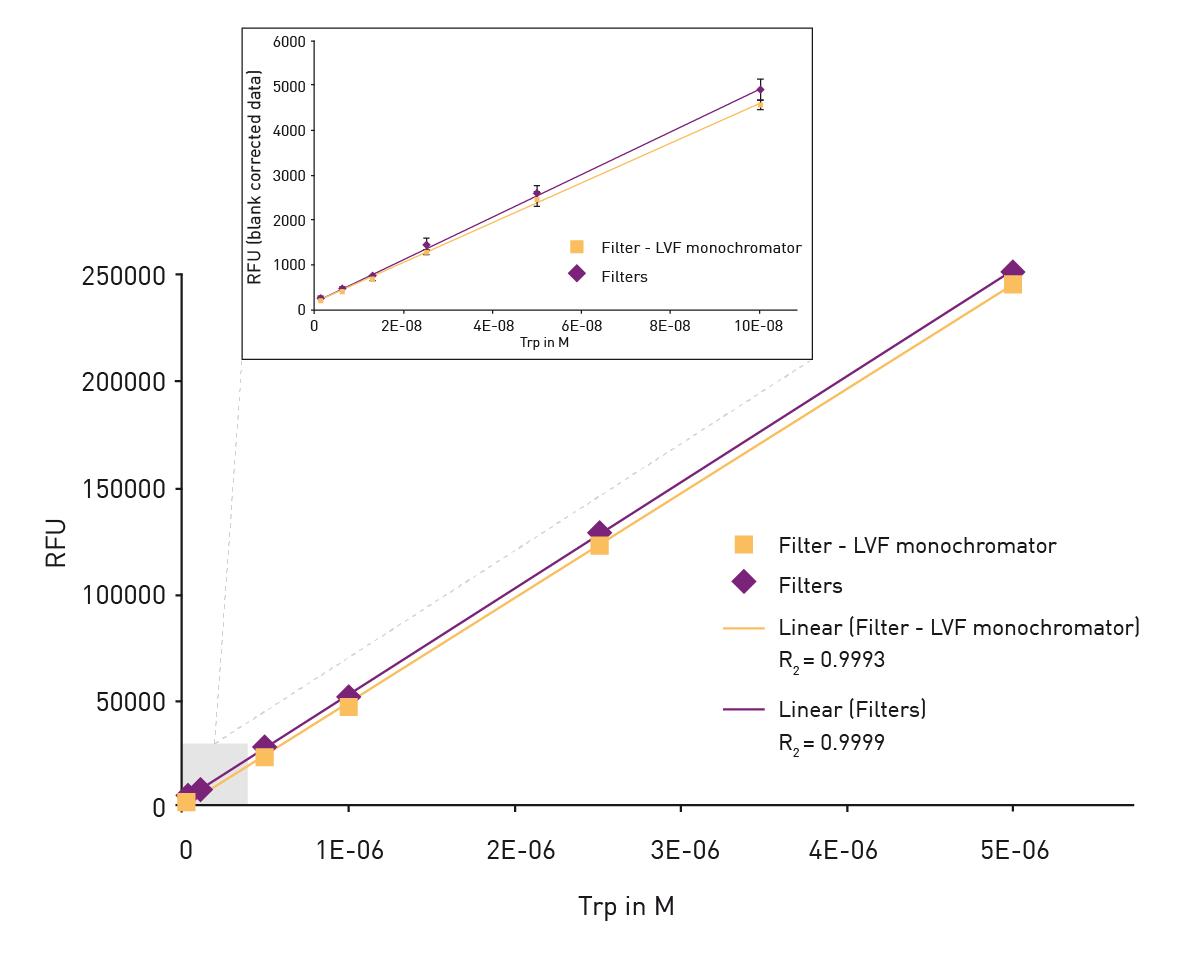
Intrinsic tryptophan detection with the SpectraMax i3 Multi-Mode Microplate Platform | Molecular Devices
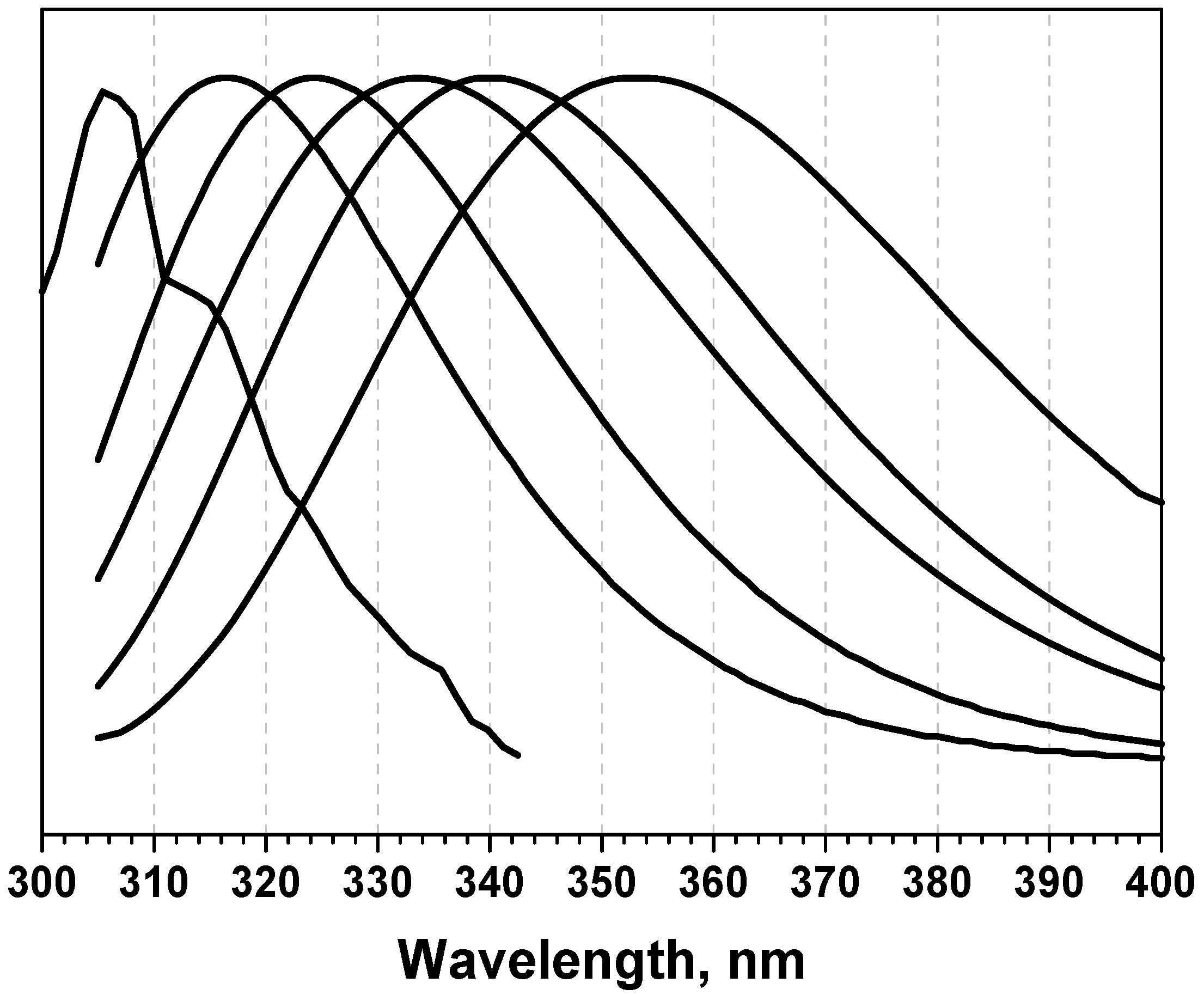
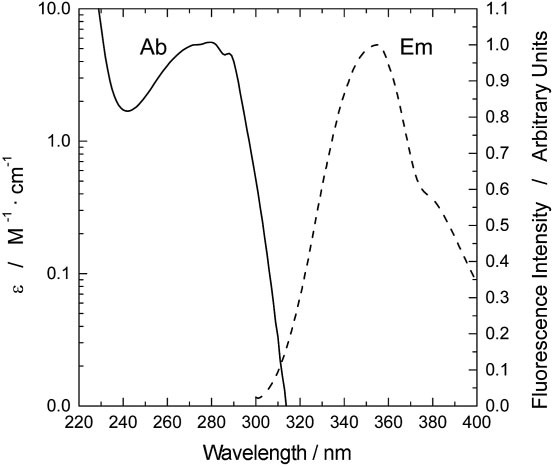
Algorithms | Free Full-Text | Algorithm for the Analysis of Tryptophan Fluorescence Spectra and Their Correlation with Protein Structural Parameters

Using tryptophan fluorescence to measure the stability of membrane proteins folded in liposomes. - Abstract - Europe PMC
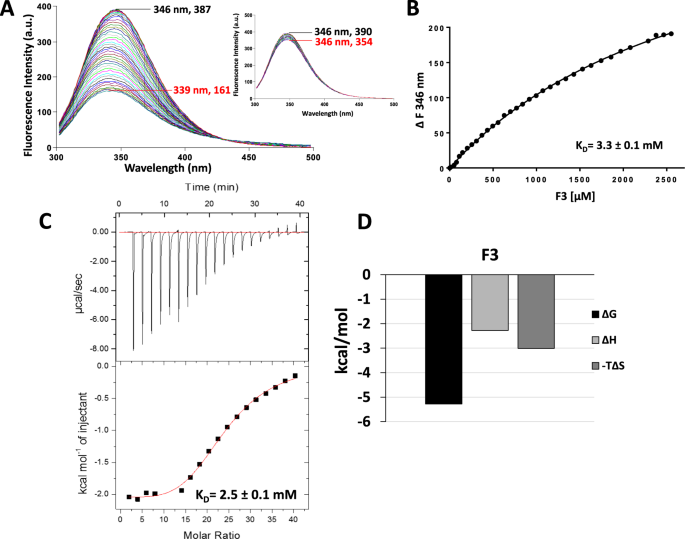
Intrinsic tryptophan fluorescence spectroscopy reliably determines galectin-ligand interactions | Scientific Reports

On the purported “backbone fluorescence” in protein three-dimensional fluorescence spectra - RSC Advances (RSC Publishing) DOI:10.1039/C6RA23426G

Intrinsic tryptophan fluorescence spectra of apoform and holoform of... | Download Scientific Diagram
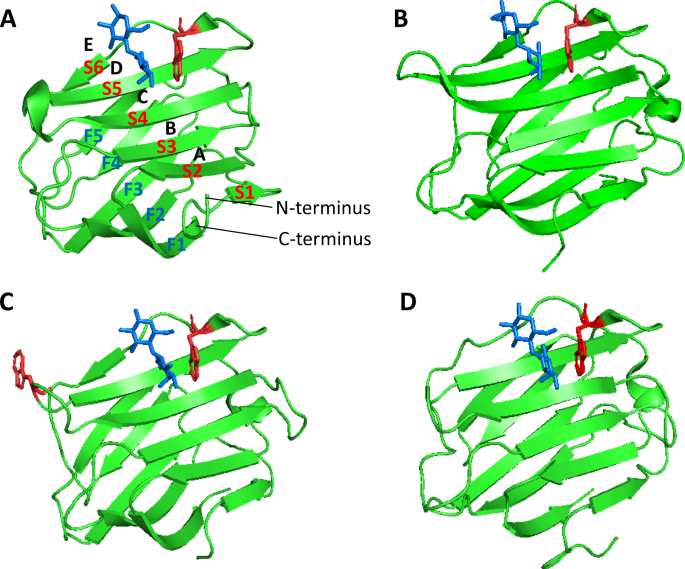
Intrinsic tryptophan fluorescence spectroscopy reliably determines galectin-ligand interactions | Scientific Reports

Intrinsic tryptophan fluorescence spectra of wild type and mutant (R12A) Hsp27, αA- and αB-crystallin.

Correlation of Tryptophan Fluorescence Spectral Shifts and Lifetimes Arising Directly from Heterogeneous Environment | The Journal of Physical Chemistry B

Peptidoglycan glycosyltransferase-ligand binding assay based on tryptophan fluorescence quenching - ScienceDirect

Quenching of tryptophan fluorescence of in situ assembled Zn2+-Peptide complex of peptide B2 (20 µM) in Tris-HCl buffer(pH 7.5, 20 mM) at 25°C on progressive titration with increasing pNPP concentration (0–400 µM) (
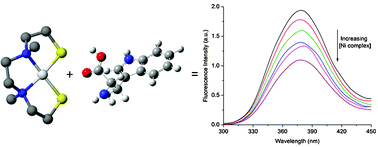
Quenching of tryptophan fluorescence in various proteins by a series of small nickel complexes - Dalton Transactions (RSC Publishing)

Trp fluorescence emission spectra. The protein samples were excited at... | Download Scientific Diagram