Multiple wireframe fractures (arrows) in a 44 mm GCA device at the 6... | Download Scientific Diagram

FDA Approves GORE CARDIOFORM Septal Occluder For PFO Closure To Prevent Recurrent Ischemic Stroke - Medical Design and Outsourcing
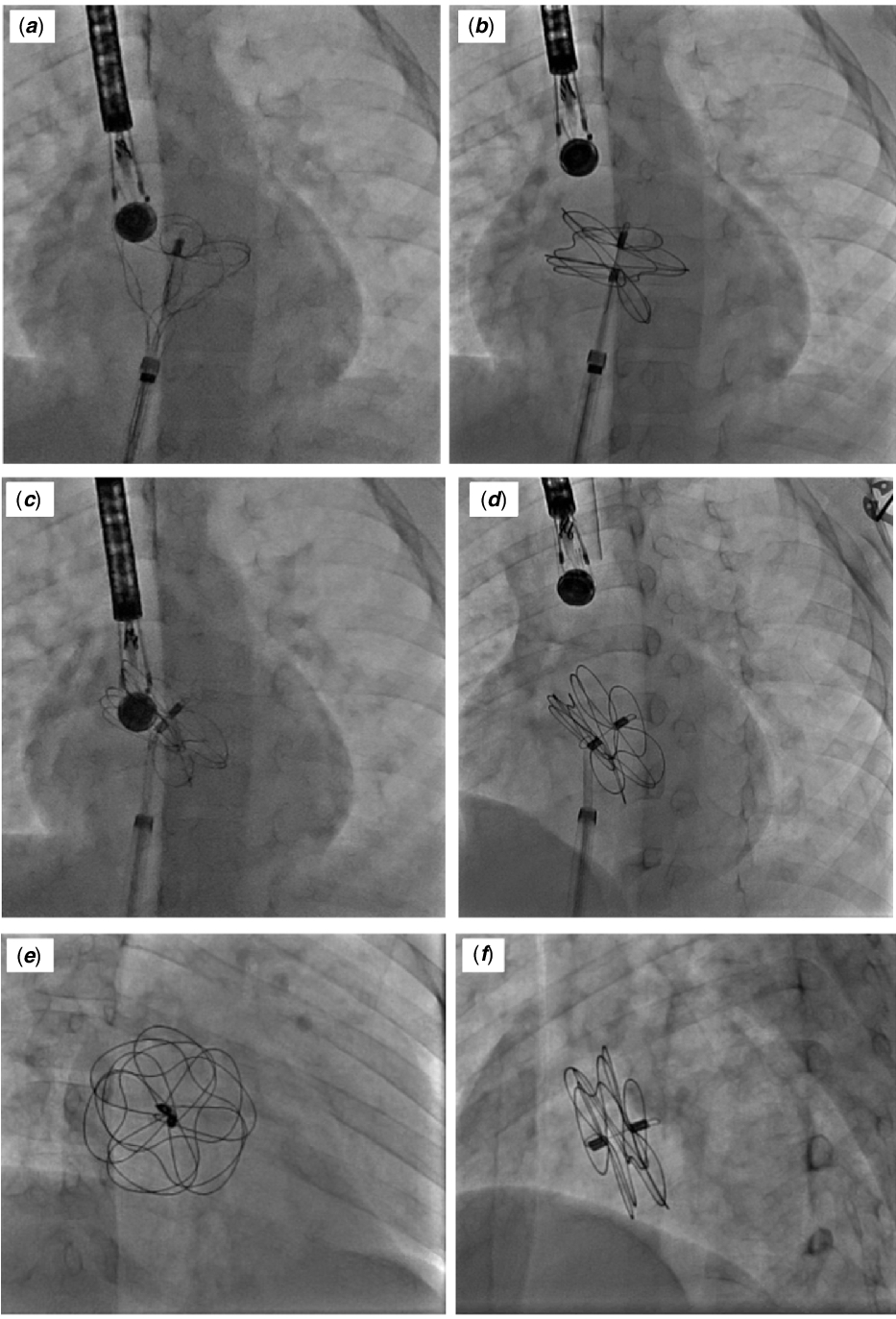
Gore Cardioform atrial septal occluder: deployment procedure and techniques for closing challenging secundum atrial septal defects | Cardiology in the Young | Cambridge Core

Les résultats à cinq ans de l'étude clinique REDUCE de Gore démontrent que la fermeture du FOP à l'aide de l'obturateur septal GORE® CARDIOFORM permet de réduire de manière sûre et à

Trans-Septal Puncture Through Gore® Cardioform Septal Occluder Device - Step by Step Approach - ScienceDirect
W. L. GORE & ASSOCIATES: GORE® CARDIOFORM ASD Occluder Receives FDA Approval for the Treatment of Atrial Septal Defects | FDA Reporter

Available Gore devices include (A) Gore Cardioform ASD Occluder, (B)... | Download Scientific Diagram

Mullins-Sheath Facilitated Delivery of Gore Cardioform ASD Occluder Devices for Closure of Large or Challenging Secundum Atrial Septal Defects

FDA Approves Gore Cardioform Septal Occluder for PFO Closure Prevent Recurrent Ischemic Stroke | DAIC

FDA Approves Gore's CARDIOFORM ASD Occluder To Treat Atrial Septal Defects | Medical Product Outsourcing

Deploy, Lock, Release. GORE® CARDIOFORM Septal Occluder device basics and closing simple PFO cases - YouTube